
Vitamin E: An Essential Nutrient for Horses
Vitamin E is an essential nutrient for horses and is beneficial in combating the many effects of free radical production that can damage membranes and components of cells. As such, vitamin E appears to be most beneficial to young rapidly growing foals, pregnant mares, stallions, and especially equine athletes. Natural and synthetic sources of vitamin E, unlike other vitamins, have different structures, and the natural form is transported quickly and retained in tissues approximately twice as long as that of synthetic vitamin E.
Introduction
Vitamin E (alpha-tocopherol) is a critically important nutrient for all horses, and supplementation is especially important for horses with limited or no access to lush pastures. This vitamin is not synthesized by the horse; therefore, it is an essential dietary nutrient. It is the primary lipid-soluble antioxidant that maintains cell membrane integrity and enhances humoral- and cell-mediated immunity. Other metabolic roles of vitamin E have been reviewed by Brigelius-Flohe and Traber (1999).
Changes in husbandry practices and ingredients used to formulate equine diets have dramatically increased the need for supplementing diets with this critically important vitamin in all segments of the horse industry. The first NRC for horses was published in 1949 and the most recent revision was published in 1989. The second revision published in 1961 devoted only one paragraph to vitamin E and concluded that “dietary supplementation of vitamin E has not been shown unequivocally to be beneficial for either prevention of reproductive troubles or muscular dystrophy in the horse” (NRC, 1961). The 1989 revision reviewed studies in horses and published minimum requirements for horses ranging from 50 to 80 IU per kg dry matter (DM) (NRC, 1989). Gestating and lactating mares, young growing horses, and performance horses have the greatest need for vitamin E supplementation, especially those that do not have access to lush, green pasture.
Free Radicals May Harm Cells
Free radicals or reactive oxygen species (ROS) are unstable atoms with unpaired numbers of electrons that are formed when oxygen interacts with other molecules in all cells. Once formed, these reactive radicals can initiate chain reactions resulting in a cascading negative effect on many other molecules within cells and cell walls, which in turn causes oxidative stress within the animal. Free radicals are commonly produced as part of normal cell metabolism, but also can become excessive following injury or disease. Left uncontrolled, free radicals can cause considerable irreparable damage to cells and cell membranes. They can alter the structure of cell membranes, and create havoc to polyunsaturated fatty acids (PUFA), proteins, and DNA within cells. The more active the cell, the greater the potential risk of cellular damage. Excessive free radical production or oxidative stress results when the formation of free radicals overwhelms the body’s ability to break the chain reactions that take place and an imbalance between production and removal of free radicals occurs. Uncontrolled oxidative stress can overpower the horse’s ability to fight back and may result in tissue damage, thus possibly impairing life.
In several species, including humans, this damage has recently been linked to degenerative diseases such as rheumatoid arthritis, cancer, cardiovascular disease, inflammatory bowel disease, renal disease, Parkinson’s disease, and cataracts. It may have a deleterious effect on the immune system (NRC Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium and Carotenoids, 2000).
Antioxidants Help Prevent Cell Damage Caused by Oxidative Stress
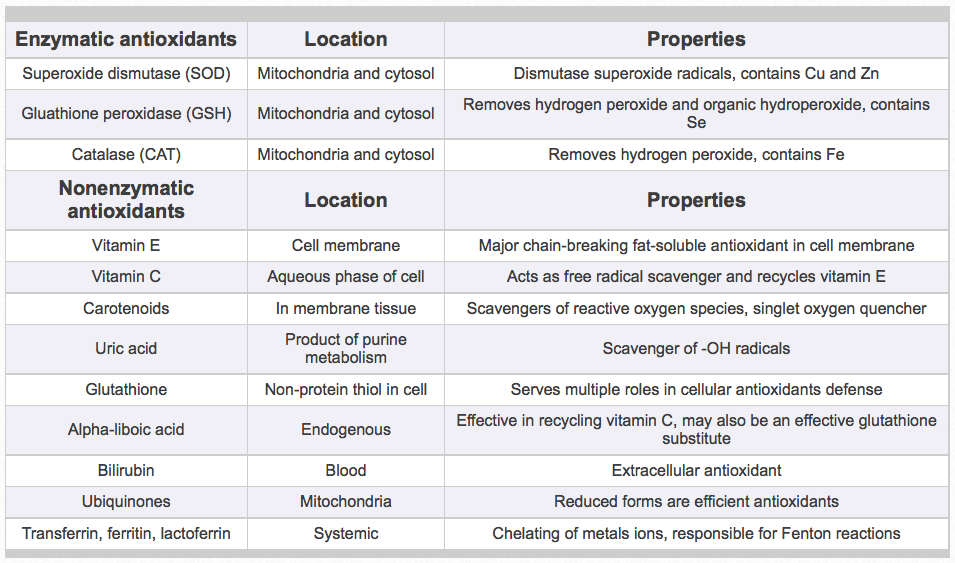
Antioxidants are the horse’s major defense system against the scourge of free radicals and oxidative stress. Enzymatic antioxidants are synthesized in the body to neutralize free radical production. Key enzymatic antioxidants include superoxide dismutase, glutathione peroxidase, and catalase (Table 1). Other major sources of antioxidants available to the horse are nonenzymatic or nutritional antioxidants. Nonenzymatic antioxidants, like vitamin E, carotenoids, vitamin C, and others, scavenge and convert free radicals to relatively stable compounds and stop the chain reaction of free radical damage (Table 1). Therefore, all antioxidants are critically important to protect horses from tissue damage and disease, and may enhance immunity during these processes. The critical phases of reproduction in mares and stallions, growth of foals, and exercise of equine athletes are all especially important. Thus, for the horse, vitamin E appears to be the most important dietary fat-soluble nonenzymatic antioxidant to assist in combating free radical production and propagation. Because horses synthesize vitamin C, vitamin E and possibly carotenoids appear to be the major antioxidant vitamins required from dietary sources.
Table 1. Important enzymatic and nonenzymatic antioxidants
Vitamin E is unique among vitamins in that it is not required for a specific metabolic function. As alpha-tocopherol, vitamin E’s major function appears to be the body’s major fat-soluble antioxidant. Thus, vitamin E is notably essential for the proper function of the reproductive, muscular, nervous, circulatory, and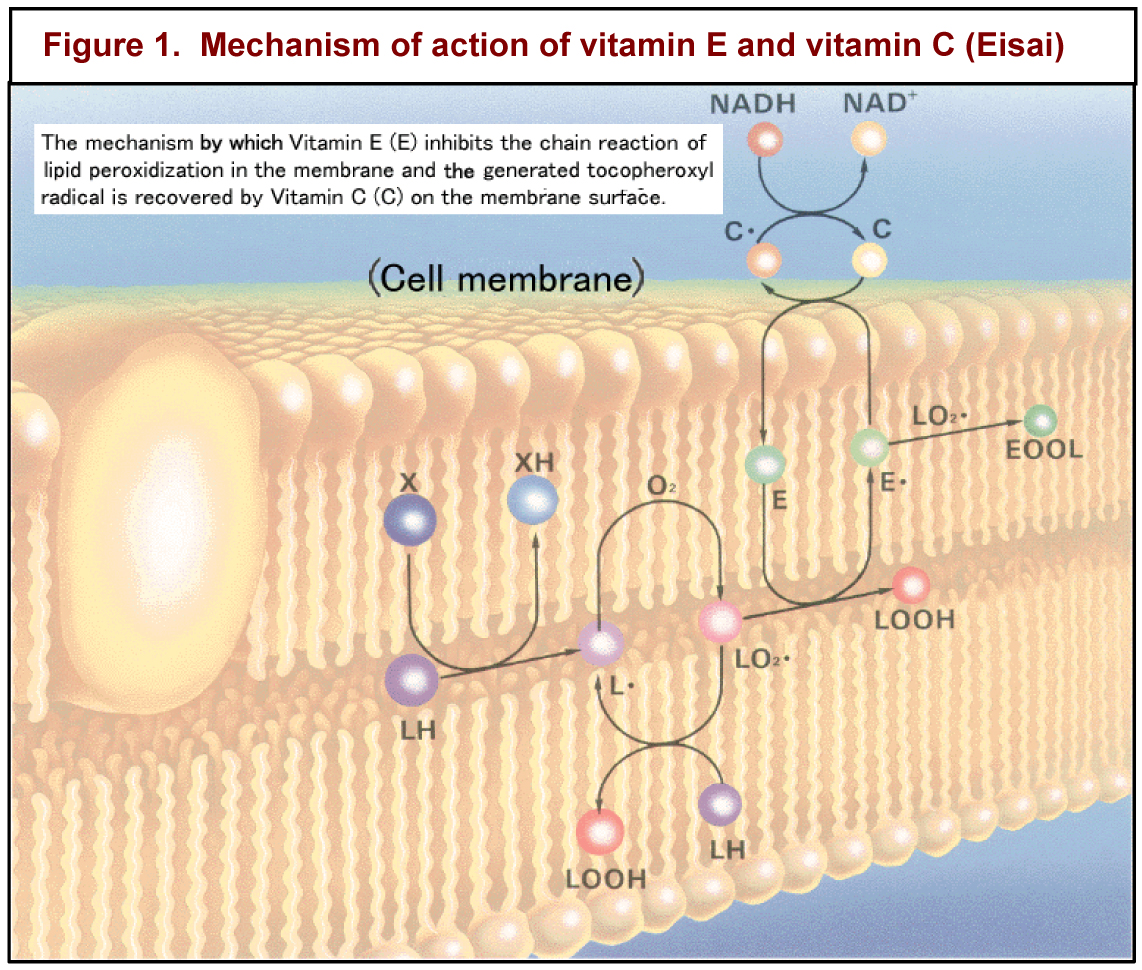 immune systems. Figure 1 shows how vitamin E and vitamin C work in concert to protect cell membranes from being oxidized. Since selenium is in glutathione peroxidase, an enzymatic antioxidant, it is often difficult to distinguish between the signs of vitamin E and selenium deficiencies.
immune systems. Figure 1 shows how vitamin E and vitamin C work in concert to protect cell membranes from being oxidized. Since selenium is in glutathione peroxidase, an enzymatic antioxidant, it is often difficult to distinguish between the signs of vitamin E and selenium deficiencies.
Vitamin E Deficiency Symptoms
Signs representing possible deficiencies of vitamin E have been extensively described in the foal and in adult horses (Schougaard et al., 1972; Wilson et al., 1976; NRC, 1989). Muscle degeneration was the most common symptom affecting both skeletal and cardiac muscle. Tongue muscles may also be affected. The latter defect may interfere with normal nursing. Steatitis has also been reported.
Vitamin E status is determined by measuring plasma or serum alpha-tocopherol levels. Low blood levels of alpha-tocopherol appear to be the first indication of vitamin E deficiencies in horses. Serum alpha-tocopherol levels above 4 µg/ml appear to be adequate for horses and levels below 2 µg/ml appear to be deficient (Schryver and Hintz, 1983). Prolonged low serum levels will lead to clinical deficiency symptoms. In order to assess vitamin E status in horses, serum or plasma alpha-tocopherol levels can be measured by HPLC.
Liu et al. (1983) reported degenerative myelopathy (spinal cord degeneration) in six Przewalski horses up to 14 years of age maintained in a zoo. They had been fed commercial horse pellets containing 22 IU vitamin E and 0.3 mg selenium/kg, timothy hay, and fresh grass in the summer. Plasma alpha-tocopherol concentrations were low and ranged from less than 0.3 to 0.8 µg/ml. Ataxia (incoordination) was evident in all, including abnormal movement of the hind limbs and abnormal wide-based gaits and stances.
Equine degenerative myeloencephalopathy (EDM) is a diffuse degenerative disease of the brain and spinal cord, a form of wobbler syndrome. Young horses affected by EDM first show signs of incoordination and clumsiness, which deteriorates over time to outright ataxia. These horses also showed low vitamin E levels in blood. Mayhew et al. (1987) found that supplementing mares with 1,500 IU vitamin E per day decreased the incidence of EDM in foals born the following year from 40% to 10%. The researchers also found that horses from 3 to 30 months of age responded to vitamin E supplementation. The mean serum alpha-tocopherol concentration of 13 ataxic weanlings was 0.62 ± 0.13 µg/ml (range 0.47 to 0.84). A number of nonataxic weanlings had similar serum values, although vitamin E supplementation markedly reduced the incidence of the syndrome on affected farms. Based on these observations, horses prone to EDM due to genetics responded to vitamin E supplementation. Blythe and Craig (1993) found that young foals showing signs of incoordination and supplemented with 6,000 IU vitamin E per day appeared normal by two years of age.
Clinical deficiency symptoms have been reported primarily in horses with limited vitamin E intake. However, subclinical vitamin E deficiencies most often go unrecognized in horses. Symptoms such as an impaired immune system or reproduction problems may often go undiagnosed and may be attributed to other causes besides inadequate vitamin E supplementation.
Determining Vitamin E Availability and Requirements
Vitamin E (alpha-tocopherol) is abundant in lush, green pastures (45 to 400 IU/kg DM), particularly in alfalfa (McDowell, 1989), and diminishes with maturation. Harvesting and length of storage diminish the quantity of vitamin E about tenfold below levels in fresh forages (Schingoethe et al., 1978). Vitamin E is abundant in the germ of grains and oils pressed from the germ (wheat germ oil, 1,330 IU/kg). Vegetable oils, such as corn and soybean oil, are relatively high in vitamin E (50-300 IU/kg), but also contain higher levels of gamma- and delta-tocopherol, that have little or no vitamin E activity. Due to such variability in vitamin E content in processed feedstuffs, nutritionists typically do not rely on the diet to provide any vitamin E activity and depend totally upon supplementation to meet horse requirements. Though horses grazing lush pasture may obtain sufficient vitamin E, it is common practice to supplement horse feeds with vitamin E at the NRC recommended levels. This is especially important for mares and working horses maintained in confinement and consuming stored roughages.
In one of the first vitamin E studies in horses, Stowe (1968) found that foals deficient in vitamin E required 27 µg of parenteral (intramuscular) or 233 µg of oral alpha-tocopherol/kg of body weight/day to maintain erythrocyte stability. Prior to HPLC methodology, erythrocyte fragility was a means to measure cell wall integrity and indirectly vitamin E status since vitamin E helps maintain cell wall integrity.
Roneus et al. (1986) suggested that vitamin E supplements be provided daily and concluded that to ensure nutritional adequacy, adult Standardbred horses fed a diet low in vitamin E should receive a daily oral supplement of 600 to 1,800 IU synthetic vitamin E acetate, equivalent to 1.5 to 4.4 IU/kg bodyweight.
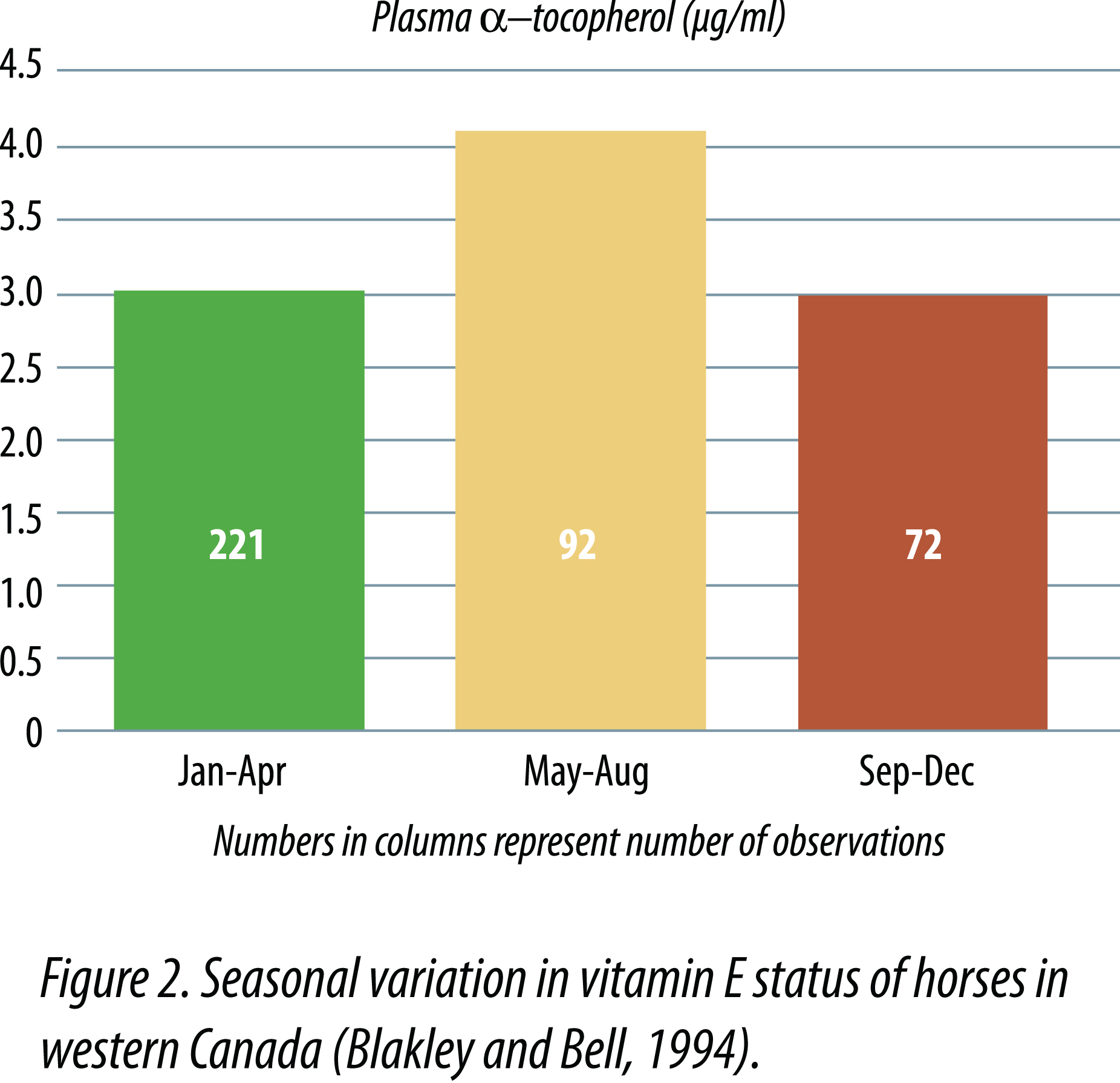
Mäenpää et al. (1988) observed seasonal differences in serum alpha-tocopherol concentrations in mares and foals kept on pasture from early June until early October, then fed timothy hay and oats through the winter. Serum alpha-tocopherol concentrations were highest in August and September (2.7 µg/ml for mares and 2.1 µg/ml for foals) and lowest in April or May (1.5 µg/ml for mares and 1.2 µg/ml for foals) after being fed hay during the winter months. When mares were given winter supplements of 100 to 400 IU of vitamin E/day, no significant increases in serumalpha-tocopherol concentrations occurred and seasonal differences persisted. However, when foals were supplemented with 400 IU vitamin E daily during winter months, serum alpha-tocopherol concentrations increased from 1.3 µg/ml to approximately 2 µg/ml in April and May. Blakley and Bell (1994) observed similar seasonal variation in plasma vitamin E (Figure 2). Janssen and Ullrey (1986) observed that the capture and restraint of zebra and Przewalski horses for hoof trimming resulted in temporary to persistent muscle soreness and lameness. They found that plasma alpha-tocopherol concentrations were commonly below 1 µg/ml in animals consuming diets supplemented with 50 IU vitamin E/kg DM. When supplemental levels were increased to 100 IU/kg DM, no signs of muscle pathology were seen, and plasma alpha-tocopherol concentrations ranged from 1.5 to 3 µg/ml after increasing vitamin E supplemental levels.
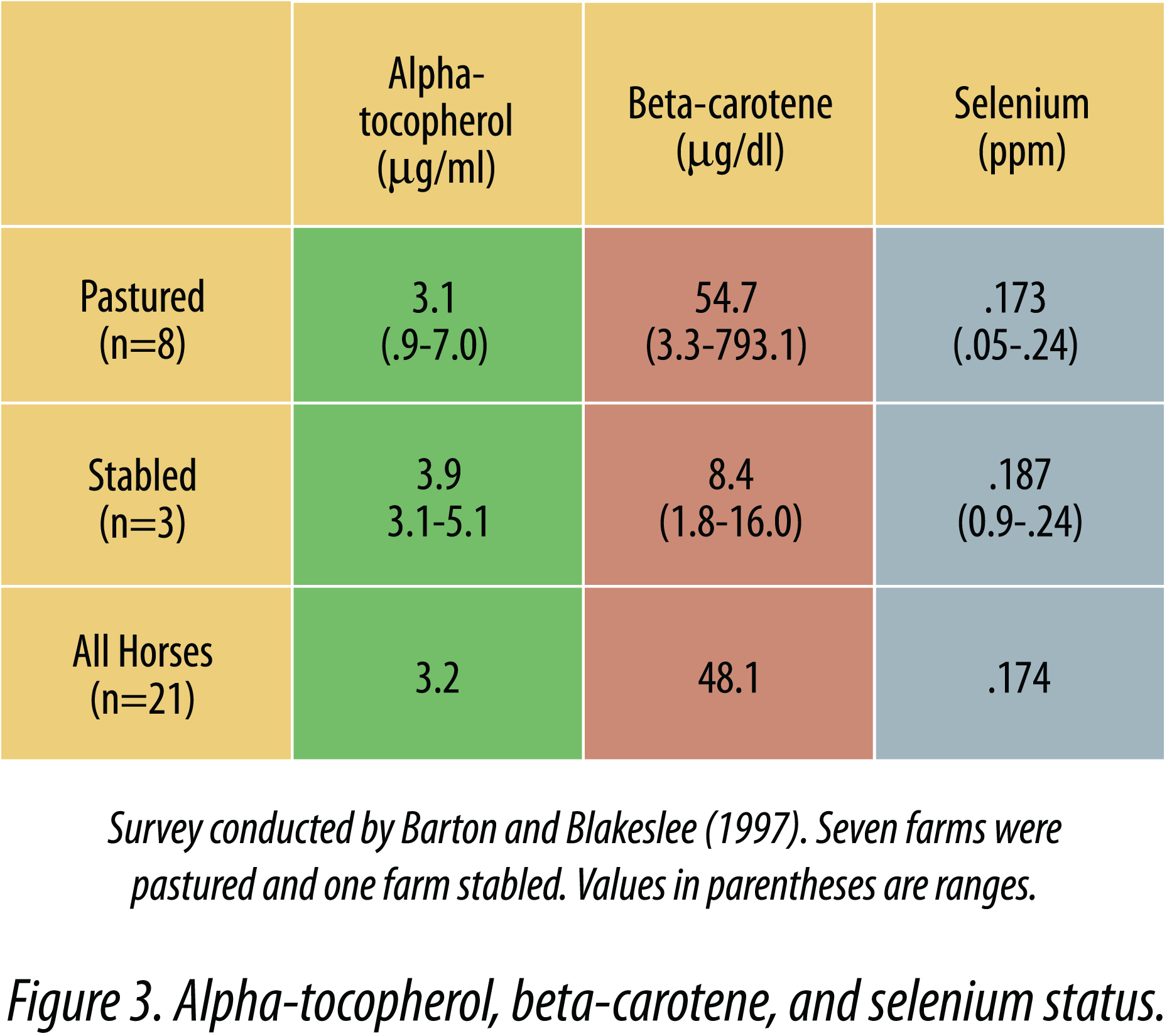
Butler and Blackmore (1983) reported that the mean plasma alpha-tocopherol concentration for 140 samples from stabled Thoroughbreds in training was 3.3 ± 1.29 µg/ml. Barton (1997) measured alphatocopherol, beta-carotene, and selenium status in horses in confinement and pasture. Serum tocopherol values ranged from 0.9 to 7.0 µg/ml in horses on pasture (Figure 3), and the range was not as great in confined horses (3.1 to 5.1). Beta-carotene levels varied dramatically in horses on grass and the average beta-carotene was much lower in confined horses. These results obtained in pastured horses are similar to results obtained by Maylin et al. (1980), who reported a range of 1.7 to 9.5 µg/ml in pastured horses.
The NRC maintenance requirement for vitamin E is 50 IU per kg DM or 0.75- 1.0 IU per kilogram body weight (1989). For growing horses, pregnant and lactating mares, and performance horses, the requirement is raised to 80 IU per kilogram DM. A dietary concentration of 100 IU of vitamin E per kg DM provides the equivalent of about 1.5 to 2 IU vitamin E per kg of body weight daily. Based on daily intake of 3% body weight, the NRC daily recommendations range from 375 IU for maintaining a 500-kg horse to 1,200 IU per day for a 500-kg lactating mare and 500-kg working horse.
Research in other species indicates that vitamin E requirements to prevent frank deficiency symptoms are considerably lower than those levels needed to improve immune status and prevent free radical damage. The amount of vitamin E required for optimum immune function has not been thoroughly studied in horses, but has been shown to be much higher than normally fed in other species (Nockels, 1979; Tengerdy, 1981). Baalsrud and Overnes (1986) supplemented a basal diet daily with 600 IU vitamin E, 5 mg selenium, or both. They observed an improved humoral immune response to tetanus toxoid or equine influenza virus in adult horses supplemented with selenium alone or selenium plus vitamin E. No improvement was seen with vitamin E alone, possibly due to an inadequate level being fed.
Vitamin E Benefits Horses During All Life Stages and Performance Levels
Mares and Foals
It is beneficial to provide supplemental vitamin E to breeding horses. Green pasture, generally high in vitamin E, is not always available to gestating and lactating mares that often foal during winter months. It is advantageous to feed mares in late pregnancy and early lactation higher levels of vitamin E in order to assure that vitamin E levels will be adequate in colostrum and milk. It has been suggested (Harper, 2002) that mares known to have poor-quality colostrum, or that had foals with failure of passive immunity transfer in previous years, should be supplemented with twice the vitamin E normally fed for at least a month before and after foaling. It was also recommended that pregnant mares fed lower quality hay should be given vitamin E supplementation a month before and after foaling. In a Swedish demonstration, Gard (2003) showed that gestating mares supplemented with 1,500 IU 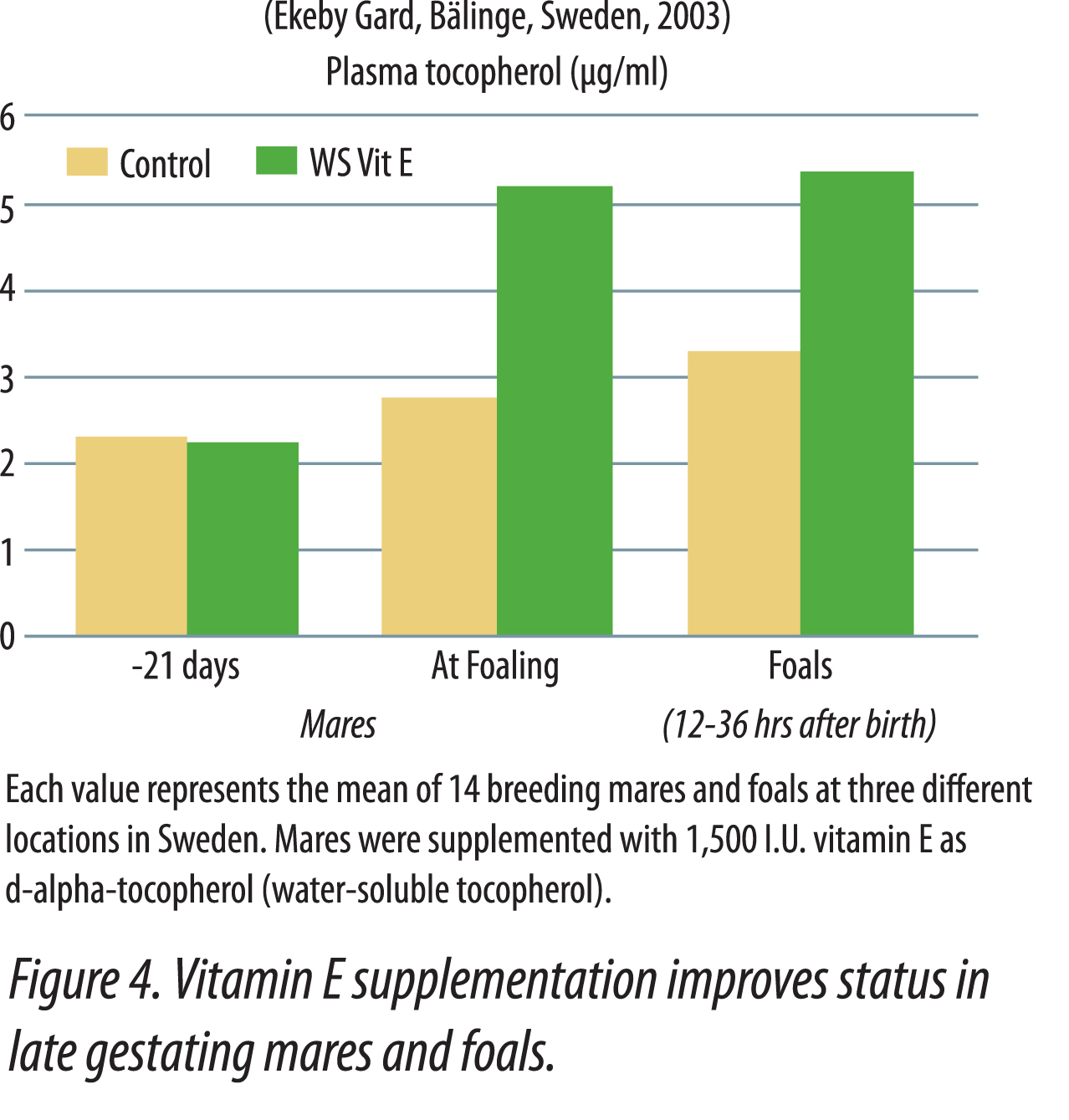 vitamin E per day as water-soluble natural vitamin E for 21 days before foaling had higher plasma alpha-tocopherol at foaling, and their foals also had higher vitamin E status at 12-36 hours after birth (Figure 4). The higher plasma tocopherol levels in foals nursing supplemented mares was attributed to higher colostrum transfer of vitamin E. Mares supplemented with vitamin E have shown increased passive transfer of antibodies to foals, which greatly enhances the immune system (Hoffman et al., 1999). This study showed an advantage of feeding vitamin E during late gestation and early lactation. Serum and colostrum IgG levels were greater in mares supplemented with 160 IU of vitamin E per kg feed (along with a mixed grass hay and grain ration), than those receiving 80 IU vitamin E per kg feed. The foals from all mares had similar levels of IgG, IgA, and IgM at birth prior to nursing. After nursing, foals from mares fed the high level of vitamin E were found to have higher serum levels of IgG and IgA, which were reflected in the dam’s colostrum.
vitamin E per day as water-soluble natural vitamin E for 21 days before foaling had higher plasma alpha-tocopherol at foaling, and their foals also had higher vitamin E status at 12-36 hours after birth (Figure 4). The higher plasma tocopherol levels in foals nursing supplemented mares was attributed to higher colostrum transfer of vitamin E. Mares supplemented with vitamin E have shown increased passive transfer of antibodies to foals, which greatly enhances the immune system (Hoffman et al., 1999). This study showed an advantage of feeding vitamin E during late gestation and early lactation. Serum and colostrum IgG levels were greater in mares supplemented with 160 IU of vitamin E per kg feed (along with a mixed grass hay and grain ration), than those receiving 80 IU vitamin E per kg feed. The foals from all mares had similar levels of IgG, IgA, and IgM at birth prior to nursing. After nursing, foals from mares fed the high level of vitamin E were found to have higher serum levels of IgG and IgA, which were reflected in the dam’s colostrum.
Exercising Horses
Decreased vitamin E status is implicated in the etiology of exercise-induced muscle damage in horses (i.e., “tying up”) (Snow & Valberg, 1994). Siciliano et al. (1997) showed that supplemental synthetic vitamin E levels of 80 IU and 300 IU per kg dry matter were necessary to maintain blood and skeletal muscle concentrations in horses undergoing exercise conditioning. Serum tocopherol levels did not affect the integrity of skeletal muscle following repeated submaximal exercise, as measured by changes in creatine kinase (CK) and aspartate amino transferase (AST) activities, nor did vitamin E level of supplementation affect thiobarbituric acid reactive substances (TBARS). Exercising horses supplemented with 2,000 IU daily for six weeks with natural vitamin E acetate (d-alpha-tocopheryl acetate) had higher plasma alpha-tocopherol levels pre- and post-exercise compared with horses receiving an equal amount of synthetic vitamin E acetate (dl-alpha-tocopheryl acetate) (Kane, 2004).
Natural Vitamin E Differs from Synthetic Vitamin E
While grazing pasture, the only source of vitamin E consumed by the horse is natural vitamin E. In nature, most plants and oils contain a mixture of tocopherols (alpha-, beta-, gamma- and delta-). Of the four tocopherols, alpha-tocopherol is the only isomer recognized to have vitamin E activity (Traber, 1999; Dietary Reference Intakes, 2000).
The two commercial sources of vitamin E are natural-source tocopherol and synthetic-source tocopherol and their corresponding acetate-esters. Synthetic vitamin sources are for the most part identical in efficacy and structure to the natural source of that vitamin. Not so for vitamin E. The source of vitamin E with the highest biological activity is natural vitamin E, which is officially recognized to have 36% greater biological activity than a synthetic source. The two sources have different structures and the natural isomer has been shown to be transported to and retained in tissues at levels approximately twice that of synthetic vitamin E (Traber et al., 1992). Natural vitamin E is one isomer (RRR-alpha-tocopherol or d-alpha-tocopherol). Synthetic vitamin E (all-rac(emic)-alpha-tocopherol or dl-alpha-tocopherol) is a racemic mixture of eight isomers, of which one or 12.5% is identical to the natural isomer and the other seven isomers are different from the natural RRR-isomer. Studies have clearly shown preferential uptake, transport, and tissue retention of the natural isomer in humans compared to synthetic sources (Ingold, et al., 1987; NRC Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium and Carotenoids, 2000). In addition, research in humans and swine has shown that milk transfer of the natural isomer is greater than for the synthetic source (Stone et al., 2003; Mahan et al., 2000).
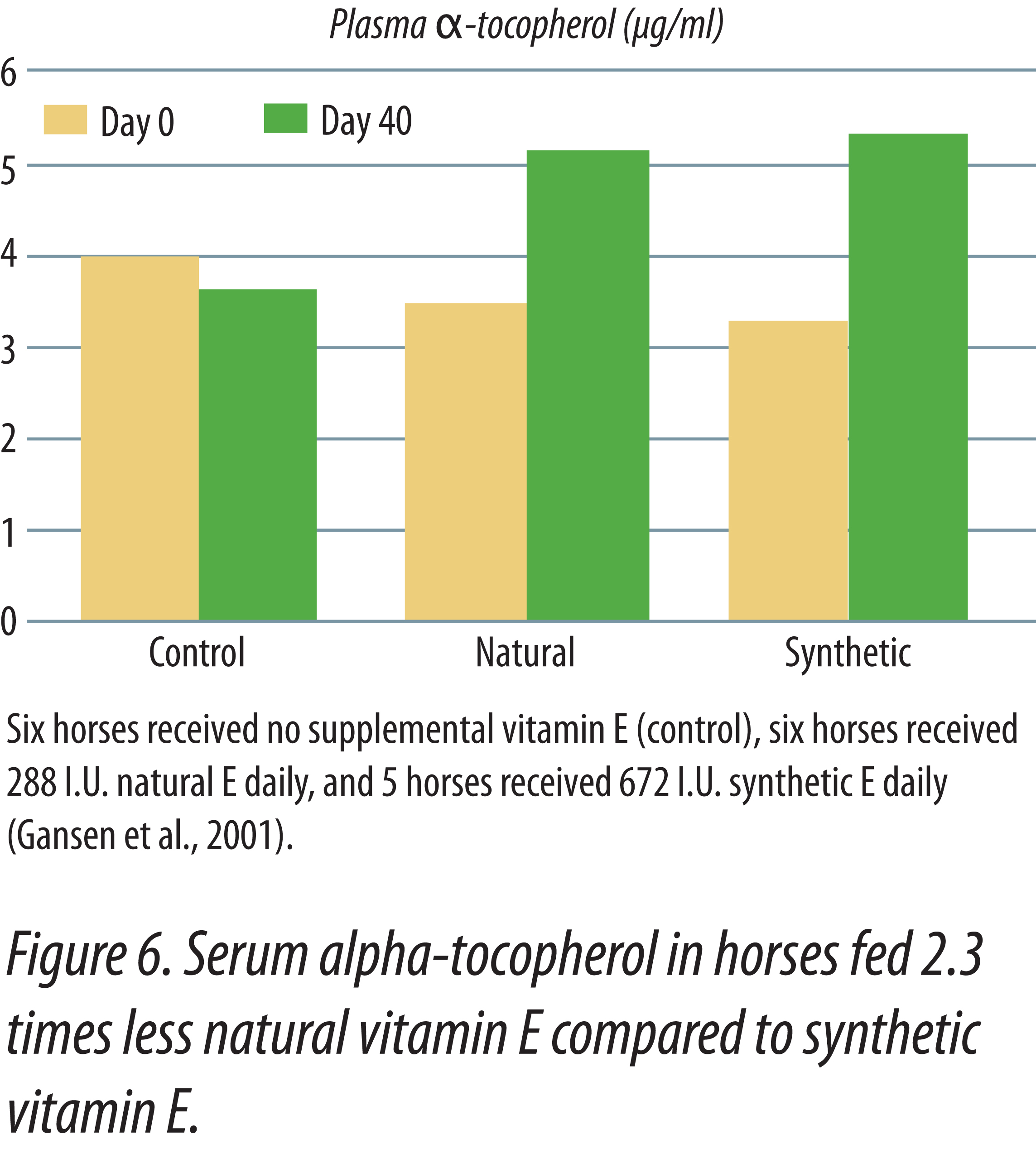
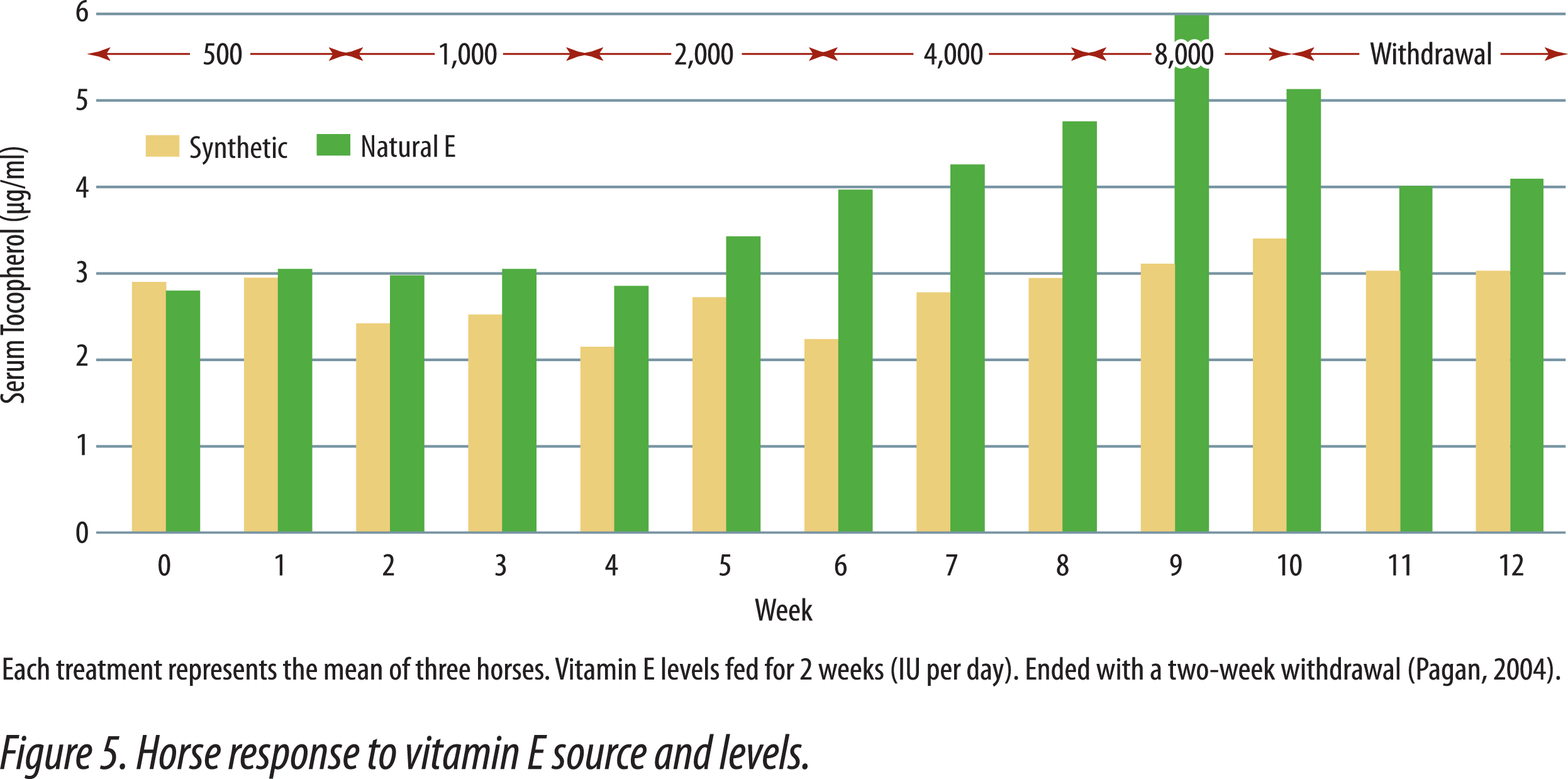
Horses fed natural vitamin E had higher plasma vitamin E levels than horses supplemented with equal IU of synthetic vitamin E (Kane, 2004). Horses were initially bled, then fed graded levels of either synthetic or natural-source vitamin E for two weeks. The initial daily supplements provided 500 IU and were doubled every two weeks until the maximum levels of 8,000 IU were fed. After two weeks at 8,000 IU, supplementation ended and the last two weeks was a depletion phase in the study. Figure 5 shows the responses obtained in the study. There was no difference in serum alpha-tocopherol when 500 IU of either natural or synthetic vitamin E was fed. As levels of supplementation increased, horses fed the natural vitamin E had progressively higher serum tocopherol levels that continued during the withdrawal period. Figure 6 shows that natural vitamin E, when fed at less than one-half the IU level of synthetic, had similar plasma tocopherol levels (Gansen et. al., 2001). These studies in horses show that the present IU values for natural and synthetic vitamin E underestimate the value of natural vitamin E. Had the published IU values been correct then there should have been no difference in blood tocopherol levels when equal amounts were fed. In human studies, natural vitamin E is recognized to have 100% the activity of an equal amount of synthetic, not 36% that was determined utilizing the rat fetal resorption assay (NRC Dietary Reference Intakes, 2000).
Importance of Form of Vitamin E-Tocopherol Utilized Better Than Tocopheryl-Ester
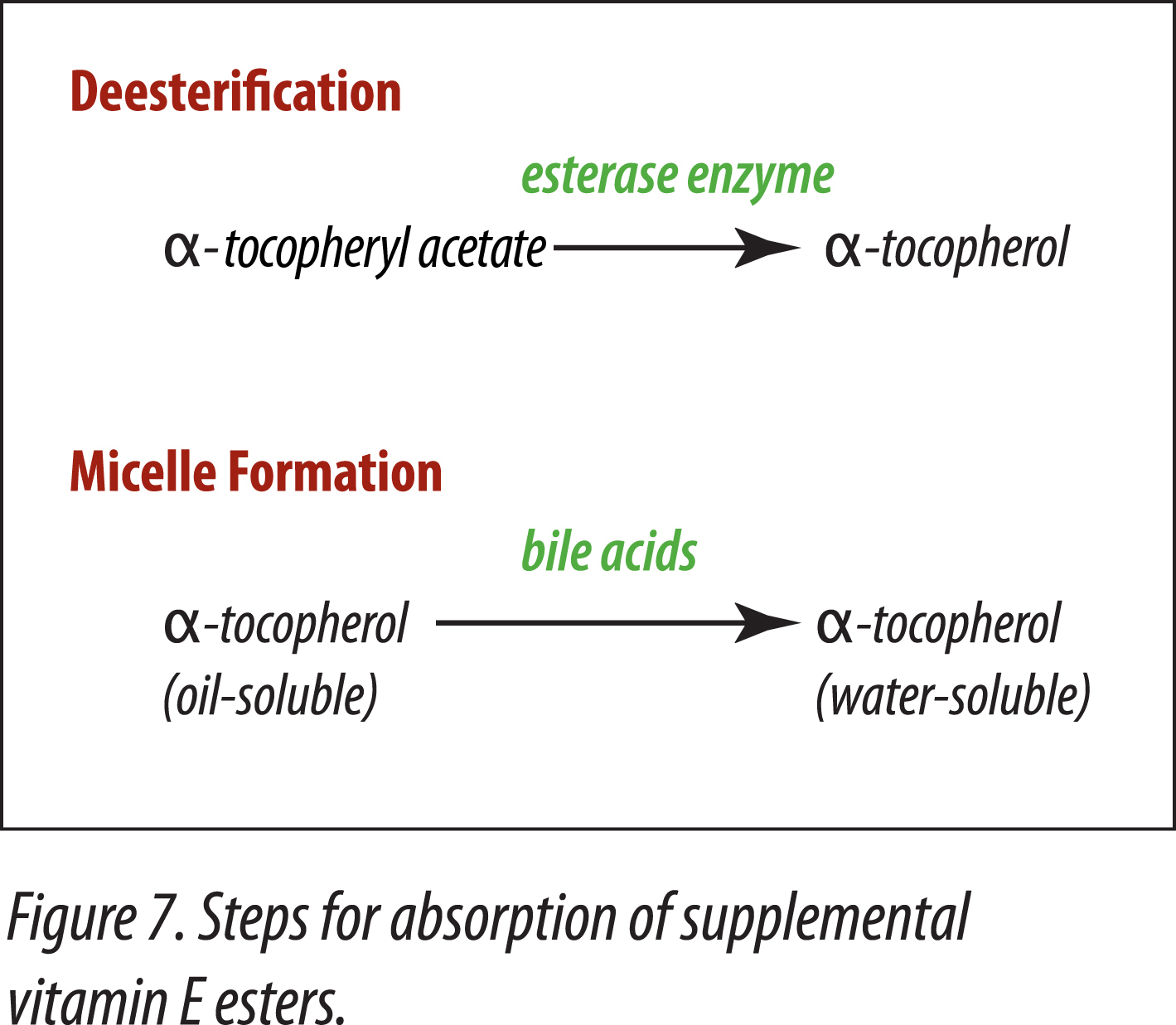
Horses must be able to efficiently absorb fat-soluble nutrients. Because natural and synthetic vitamins E are antioxidants, in order for them to be included into rations, and not lose their potency, esters are added to provide stability. In order for supplemental vitamin E-ester to be utilized, two steps are necessary. The ester has to be removed and the alpha-tocopherol has to be made water-soluble by the action of bile salts (Gallo-Torres, 1980) (Figure 7).
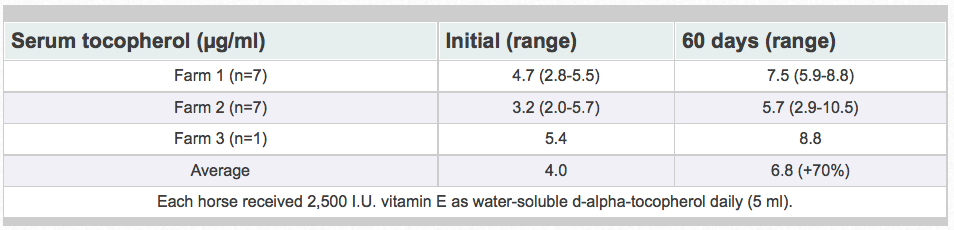
Research has shown that water-soluble, unesterifed alpha-tocopherol is better utilized than acetate-esters. Of vitamin E products tested in horses, the most efficient one was a water-soluble source of d-alpha-tocopherol. The manufacturing process creates tiny microscopic water-soluble droplets (optimally 0.1 to 0.4 microns). This process dramatically enhances the ability of horses to absorb vitamin E. Because it is water soluble, absorption is greatly enhanced. It does not require the acetate-ester to be hydrolyzed prior to absorption and there is no need for micellization by bile salts. Figure 8 shows the results when horses were fed either 0, 1,000, or 2,000 IU vitamin as RRR-alpha-tocopherol (Elevate®).Table 2 shows results when horses were fed 2,500 IU natural vitamin E. Vitamin E status was improved 70% with the supplementation of the water-soluble product. Similar results have been obtained in other animal species (data on file).
Table 2. Water-soluble tocopherol supplementation in horses.
Summary
Vitamin E is the primary lipid-soluble antioxidant that protects the cell membranes of many systems of the horse’s body. Vitamin E is 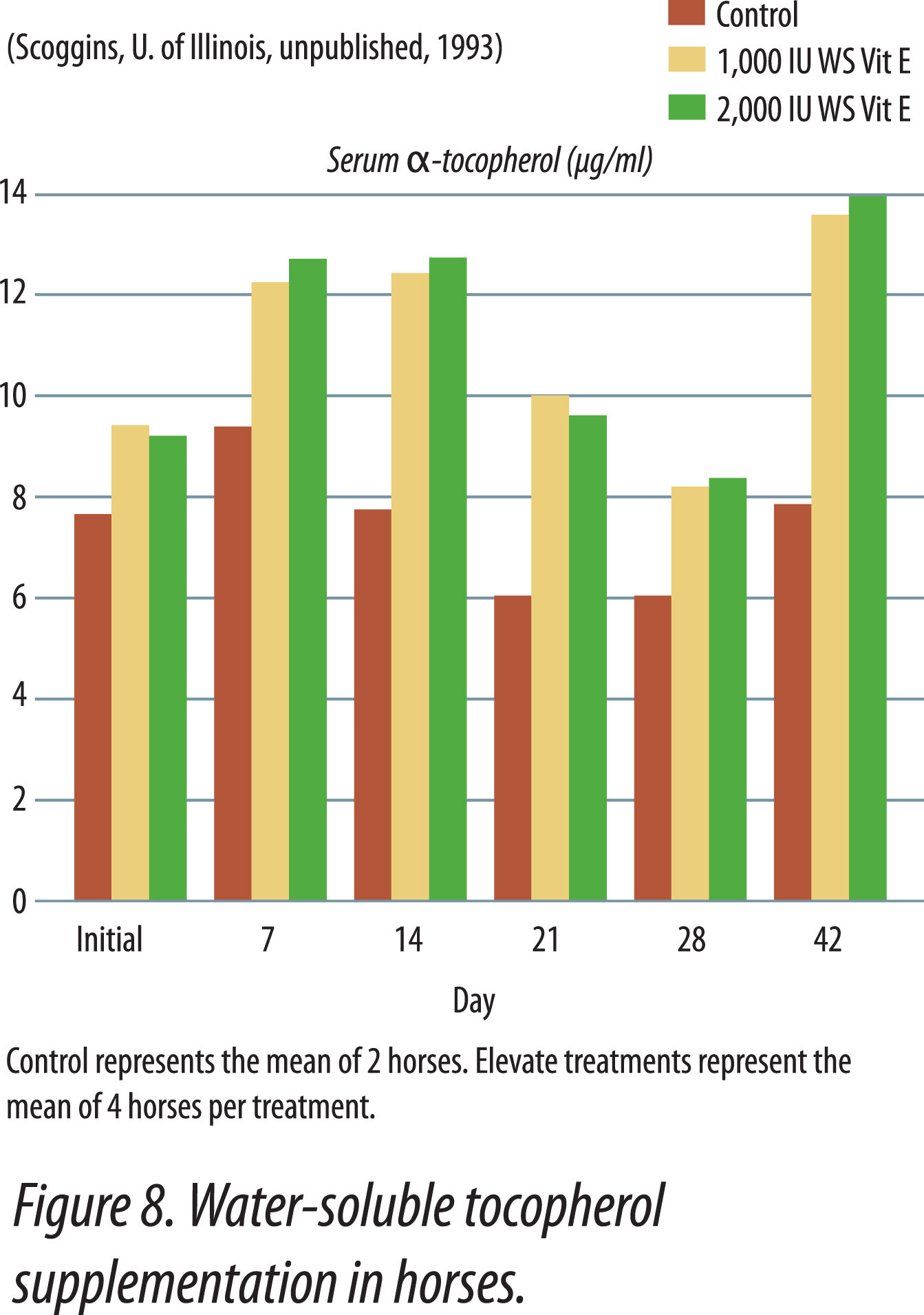 not a “typical” vitamin because the deficiencies do not always produce overt symptoms like other vitamin deficiencies. The need to supplement horses with vitamin E is primarily dependent on whether they graze lush pastures adequate in vitamin E or are kept in confinement and fed diets low in vitamin E. The requirements appear to be higher when enhanced immunity is expected, and greater in newborn foals, breeding animals, and performance horses. Not all forms and sources of vitamin E are equally utilized by horses. Natural vitamin E appears to be better utilized than synthetic.
not a “typical” vitamin because the deficiencies do not always produce overt symptoms like other vitamin deficiencies. The need to supplement horses with vitamin E is primarily dependent on whether they graze lush pastures adequate in vitamin E or are kept in confinement and fed diets low in vitamin E. The requirements appear to be higher when enhanced immunity is expected, and greater in newborn foals, breeding animals, and performance horses. Not all forms and sources of vitamin E are equally utilized by horses. Natural vitamin E appears to be better utilized than synthetic.
Recommendations
The vitamin E requirements stated in the NRC Nutrient Requirements of Horses (1989) are the minimum amounts needed to sustain normal activities in horses. The vitamin E requirements are met from naturally occurring vitamin E in feedstuffs, primarily the roughage sources, and supplemental sources in the concentrate. The NRC lists several factors that apply to all nutrient needs as well as vitamin E. Some of the considerations are interrelationships among nutrients, previous nutritional status, climatic and environmental conditions, and variation in nutrient availability in feed ingredients. Horses with minimum activity and maintained on green pasture typically do not need vitamin E supplementation due to adequate natural vitamin E in the fresh grass. However, due to loss of vitamin E in stored and processed roughages, horses in confinement need vitamin E supplementation. Table 3 gives recommended supplemental natural vitamin E levels for the various classes of horses. To achieve maximum blood and tissue levels of alpha-tocopherol, we recommend that horse feeds be supplemented with natural-source vitamin E, as RRR-alpha-tocopherol (Elevate®) as a top-dress.
Table 3. Recommended supplemental natural vitamin E for various classes of horses

Serum alpha-tocopherol levels greater than 4.0 µg/ml would indicate that the vitamin E supplementation program is adequate. In order to achieve this blood level, vitamin E supplementation may exceed 50-80 IU/kg DM, which is well below the maximum tolerance level of 1,000 IU per kg DM (NRC, 1989). More data are needed to precisely determine the proper recommendation of vitamin E for various classes of horses. Foals, pregnant and lactating mares, and high-performance working horses have the greatest need for natural-source vitamin E supplementation.
References
Baalsrud, K. J., & Overnes, G. 1986. The influence of vitamin E and selenium supplements on antibody production in horses. Eq. Vet. J. 18:472.
Blakley, B.R., & Bell, R.J. 1994. Seasonal variation of vitamin E status in horses. Can. Vet. J. 35:297.
Blythe, L.L., & Craig, A.M. 1993. Equine degenerative myeloencephalopathy. Part 1. Clinical signs and pathogenesis. Equine Compendium. 14:1215.
Butler, P., & Blackmore, D.J. 1983. Vitamin E values in the plasma of stabled Thoroughbred horses in training. Vet. Rec. 112:60.
DRI. 2000. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. National Academy Press, Wash. D.C.
Gallo-Torres, H. E. 1980. Absorption, transportation and metabolism. In: L. J. Machlin (Ed.). Vitamin E. A Comprehensive Treatise. p. 185. Marcel Dekker. New York.
Harper, F. 2002. Vitamin E for horses. Univ. Tenn. Horse Information Series, AS-H 126.
Hoffman, R.M., Morgan, K.L., Lynch, M.P., Zinn, S.A., Faustman, C., & Harris, P.A. 1999. Dietary vitamin E supplemented in the periparturient period influences immunoglobulins in equine colostrums and passive transfer in foals. In: Proc. Equine Nutr. Physiol. Soc. Symp. p. 96.
Ingold, K.U., Webb, A.C., Witter, D., Burton, G.W., Metcalfe, T. A., & Muller, D.P.R. 1987. Vitamin E remains the major lipid-soluble, chain-breaking antioxidant in human plasma even in individuals suffering severe vitamin E deficiency. Arch. Biochem. Biophys. 259:224.
Janssen, D. L., & Ullrey, D.E. 1986. Cited in NRC of Horses 1989.
Kane, E., 2004. Vitamin E is essential equine nutrient. DVM Magazine, May 2004. p. 10E-15E.
Liu, S.K., Dolensek, E.P., Adams, C.R., & Tappe, J.P. 1983. Myelopathy and vitamin E deficiency in six Mongolian wild horses. J. Am. Vet. Med. Assoc. 183:1266.
Mäenpää, P.H., Koskinen, T., & Koskinen, E. 1988a. Serum profiles of vitamins A, E, and D in mares and foals during different seasons. J. Anim. Sci. 66:1418.
Mahan, D.C., Kim, Y.Y., & Stuart, R.L. 2000. Effect of vitamin E sources (RRR- or all-rac-a-tocopherol) and levels on sow reproductive performance, serum, and milk a-tocopherol contents over a five-parity period, and the effects on the progeny. J. Anim. Sci. 78:110.
Mayhew, I.G., Brown, C.M., Stowe, H.D., Trapp, A.L., Derksen, F.J., & Clement, S.F. 1987. Equine degenerative myeloencephalopathy: A vitamin E deficiency that may be familial. J. Vet. Intern. Med. 1:45.
Maylin, G. A., Rubin, D. S. & Lein, D. H. 1980. Selenium and vitamin E in horses. Cornell Vet. 70:272.
McDowell, L.R. 1989. Vitamin E. In: Vitamins in Animal Nutrition. Comparative aspects to human nutrition. p. 20. Academic Press. San Diego, CA.
Nockels, C.F. 1979. Protective effects of supplemental vitamin E against infection. Fed. Proc. 38: 2134.
NRC. 1961. Nutrient Requirements of Horses (2nd Ed.). National Academy Press, Washington, DC.
NRC. 1989. Nutrient Requirements of Horses (5th Ed.). National Academy Press, Washington, D.C.
Roneus, B.O., Hakkarianen, R.V. J., Lindholm, C.A., & Tyopponen, J.I. 1986. Vitamin E requirements of adult Standardbred horses evaluated by tissue depletion and repletion. Eq. Vet. J. 18:50.
Schingoethe, D.J., Parsons, J.G., Ludens, F.C., Tucker, W.L., & Shave, H.J. 1978. Vitamin E status of dairy cows fed stored feeds continuously or pastured during summer. J. Dairy Sci. 61:1582.
Schougaard, H., Basse, A., Gissel-Nielsen, G., & Simesen, M.G. 1972. Nutritional muscular dystrophy (NMD) in foals. Nord. Veterinaermed. 24:67.
Schryver, H., & Hintz, H. F. 1983. Vitamins. In: N.E. Robinson (Ed.). Current Therapy in Equine Medicine. p. 84. W.B.Saunders, Philadelphia.
Siciliano, P.D., Parker, A.L., & Lawrence, L.M. 1997. Effect of dietary vitamin E supplementation on the integrity of skeletal muscle in exercised horses. J. Anim. Sci. 75:1553.
Snow, D.H., & Valberg, S.J, 1994. Muscle anatomy, physiology, and adaptations to exercise and training. In: R. J. Rose and D. R. Hodgson (Eds.) The Athletic Horse: Principles and Practice of Equine Sports Medicine. p. 145. W. B. Saunders, Philadelphia.
Stone, W.L., LeClaire, I., Ponder, T., Baggs, G., & Barrett-Reis, B. 2003. Infants discriminate between natural and synthetic vitamin E. Am. J. Clin. Nutr. 77:899.
Stowe, H.D. 1968. Alpha-tocopherol requirements for equine erythrocyte stability. Am. J. Clin. Nutr. 21:135
Tengerdy, R.P. 1981. Vitamin E, immunity and disease resistance. Adv. Exp. Med. Biol. 135: 27.
Traber, M.G. 1999. Vitamin E. In: M.E. Shils, J.A. Olson, M. Shike, and A.C. Ross (Eds). Modern Nutrition in Health and Disease (9th Ed.). p. 347. Williams & Wilkins, Baltimore.
Traber, M.G., Burton, G. W., Hughes, L., Ingold, K.U., Hidaka, H., Malloy, M., Kane, J., Hyams, J., & Kayden, H.J. 1992. Discrimination between forms of vitamin E by humans with and without genetic abnormalities of lipoprotein metabolism. J. Lipid Res. 33:1171.
Vandergriff, W. 1998. In: E is for ‘ease’. Vitamin E may ease the symptoms of motor neuron disease and other stressors. K. Marcella. Thoroughbred Times. February, 1998.
Wilson, T.M., Morrison, H.A., Palmer, N.C., Finley, G.G., & van Dreumel, A.A. 1976. Myodegeneration and suspected selenium/vitamin E deficiency in horses. J. Am. Vet. Med. Assoc. 169:213.



5 Comments
I have a 27 year old Paso fino gelding he had a hairline fracture in his back leg caused by a ferrier he is not steady on his feet had vet she suggest giving him vit E what is the best kind to give him. He cannot be ridden or worked due to his leg. So he keeps my other paso fino company and he is my baby old man for I can never ride again had 8 back operations was told never ride again so he and me are basically lawn ornaments but we love each other I spend as much time as I can with him .so if I could get the name of the best vit E to give him TIA .
Hello Terry,
Thank you for asking your question on our blog. I am sorry to hear about your Paso. He is lucky to have an owner like you who loves him so much.
The best kind of vitamin E for your gelding is Elevate® Maintenance Powder natural vitamin E. I would recommend giving him 3 scoops per day for 60 days, and if you see an improvement, you can reduce the amount fed to 2 scoops per day.
Below is a link with more information about Elevate Maintenance Powder. If you have any other questions please let me know.
https://kppusa.com/product/elevate-maintenance-powder/
Had a problem with my horse not picking up his hind feet. Had a chiropractor and my vet out and they couldn’t figure out why he had this problem. Vet took blood-found he was very deficient in Vit E. Started feeding Elevate last August, 2018. He has begun to pick up his hind feet now, in May 2019. Also, he shed his winter coat with very little help from me. He is galloping around like a two year old, and he’s 12. All my horses are on Elevate. My mare was lame, off and on, in her back legs-she is dashing around like a nut, and no lameness since started on Elevate, 10 months now. Also-all three horses were treated for Lyme Disease last year. I have seen no problems from the Lyme, and strongly suggest any one with a horse that has had Lyme, to get their horses on Vitamin E as soon as possible. And keep them on it!!!! Wish I knew about this information earlier. Wondering if it could help prevent Cushings.
Hi Karen,
Thanks so much for the positive feedback. I am so happy to hear your horses are doing so well. Regarding your question about Cushing’s, pituitary pars intermedia dysfunction (PPID) is an endocrine disease that causes the pituitary gland to malfunction. PPID is also called Cushing’s disease. The natural vitamin E in Elevate provides antioxidant and immune support to horses that are struggling with PPID, but it will not prevent the disease.
Pingback
[…] wasting. Did you see any improvement with the E? It sometimes needs to be in high doses to help. Vitamin E: An Essential Nutrient for Horses | KPP I definitely wouldn't stop giving it as long as she appears to have muscle wasting, because she […]